
HS Dosing COSENTYX® (secukinumab)

Cosentyx Dosage Guide
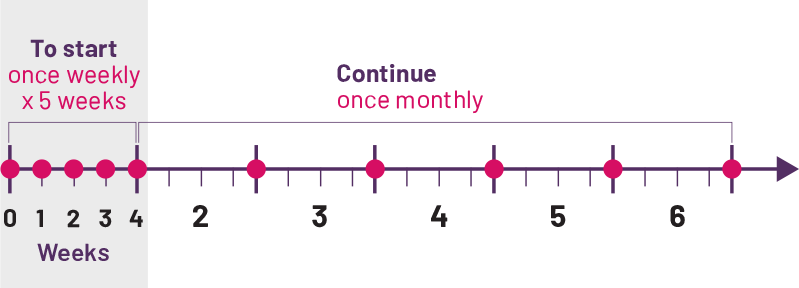
Psoriatic Arthritis Dosing

Secukinumab recommended by NICE for hidradenitis suppurativa - Hospital Pharmacy EuropeHospital Pharmacy Europe

WO2019097493A1 - Treating hidradenitis suppurativa with il-17 antagonists - Google Patents

FAQ, COSENTYX® (secukinumab)

Secukinumab (AIN457), IL-17A Antagonist

FDA Approves Novartis' Cosentyx as First New Biologic HS Treatment in Decade: A Potential Rival for AbbVie's Humira

FDA Approves Cosentyx as Treatment Option for Hidradenitis Suppurativa
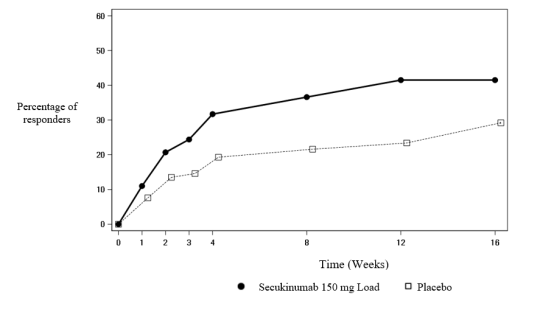
Cosentyx 150 mg solution for injection in pre-filled pen - Summary of Product Characteristics (SmPC) - (emc)

These highlights do not include all the information needed to use COSENTYX safely and effectively. See full prescribing information for COSENTYX. COSENTYX® (secukinumab) injection, for subcutaneous or intravenous useInitial U.S. Approval: 2015
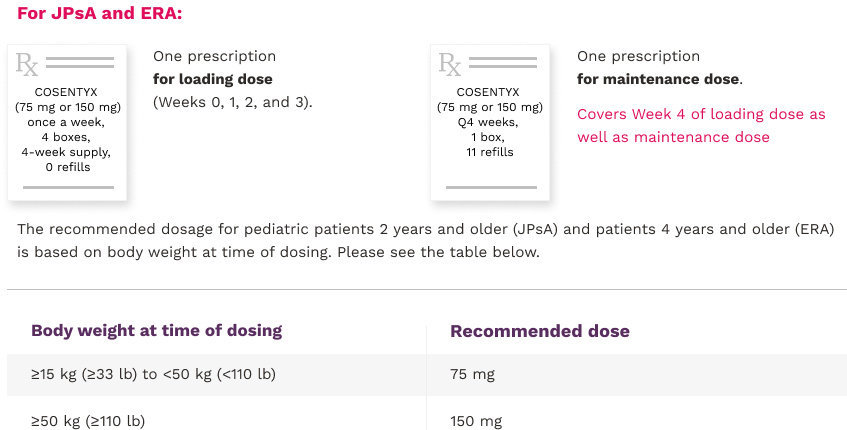
Rheumatology Dosing, COSENTYX® (secukinumab)
Cosentyx: Package Insert








/cloudfront-eu-central-1.images.arcpublishing.com/diarioas/IR2YVHF5YRBGXAU7HOJ4AHUY6E.jpg)
